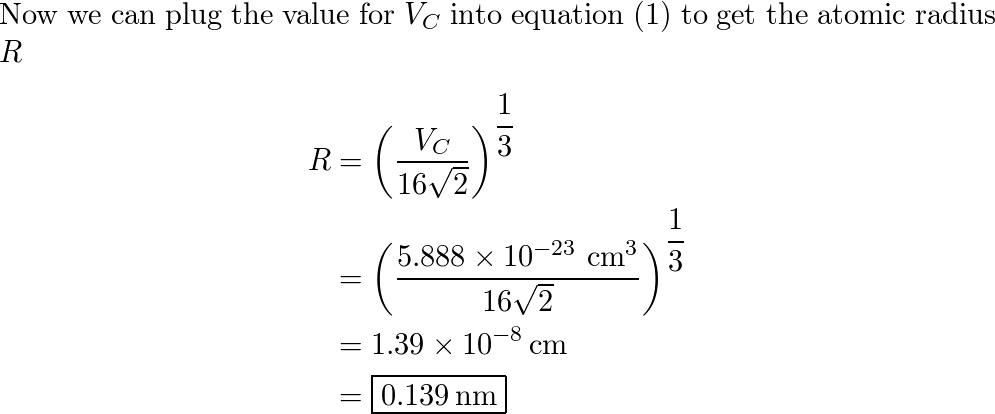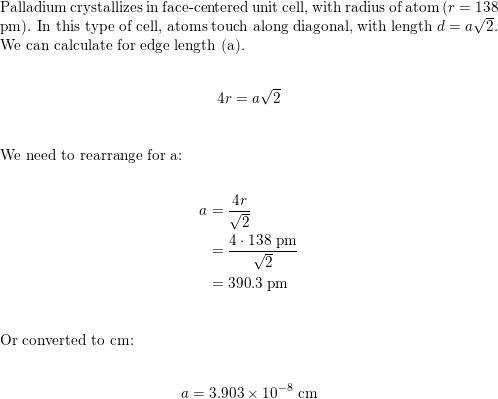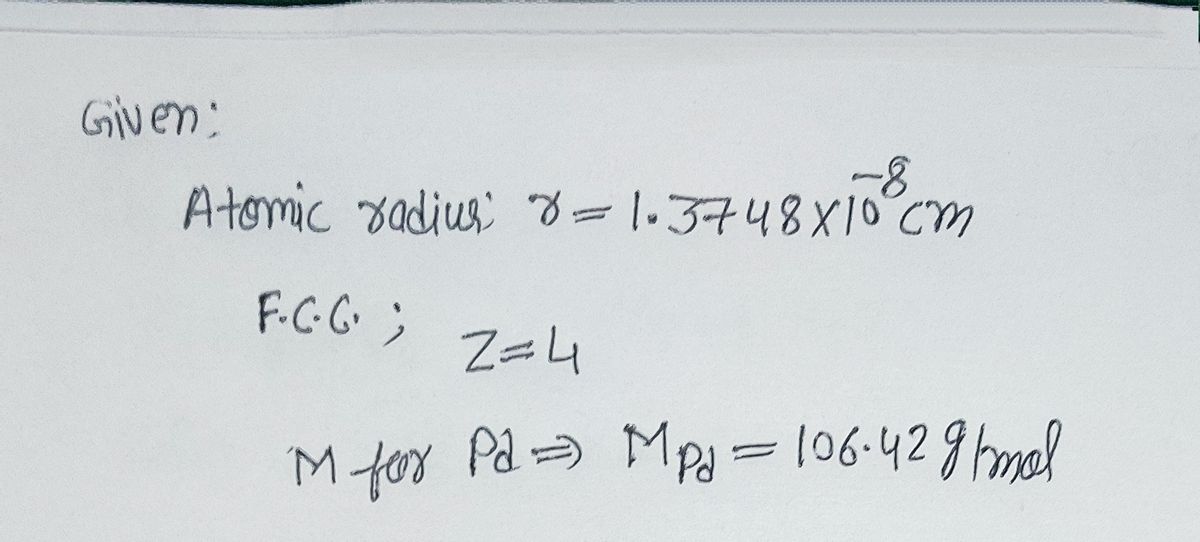OneClass: A metal crystallizes in the face-centered cubic (FCC) lattice. The density of the metal is ...
PLEASE HELP! 80 points!! A metal crystallizes in the face‑centered cubic (FCC) lattice. The density of the - Brainly.com
Metallic gold crystallizes in the facecentred cubic lattice. The length of the cubic unit cell is a = 4.070 Å. - Sarthaks eConnect | Largest Online Education Community

A Ketimide-Stabilized Palladium Nanocluster with a Hexagonal Aromatic Pd7 Core | Inorganic Chemistry

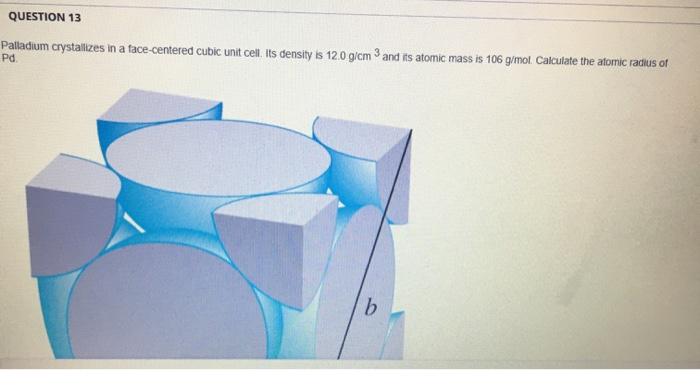
roblem.docx - problem #1: Palladium crystallizes in a face-centered cubic unit cell. Its density is 12.023 g/cm3. Calculate the atomic radius of | Course Hero
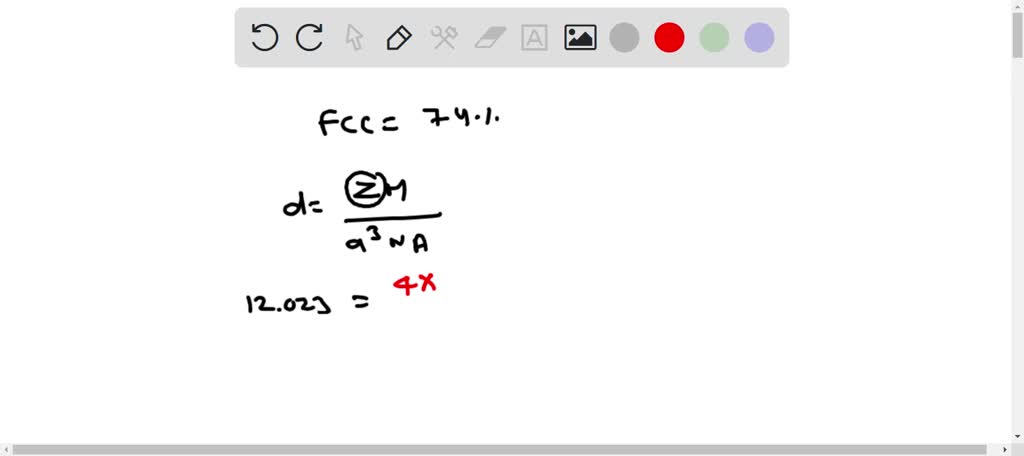
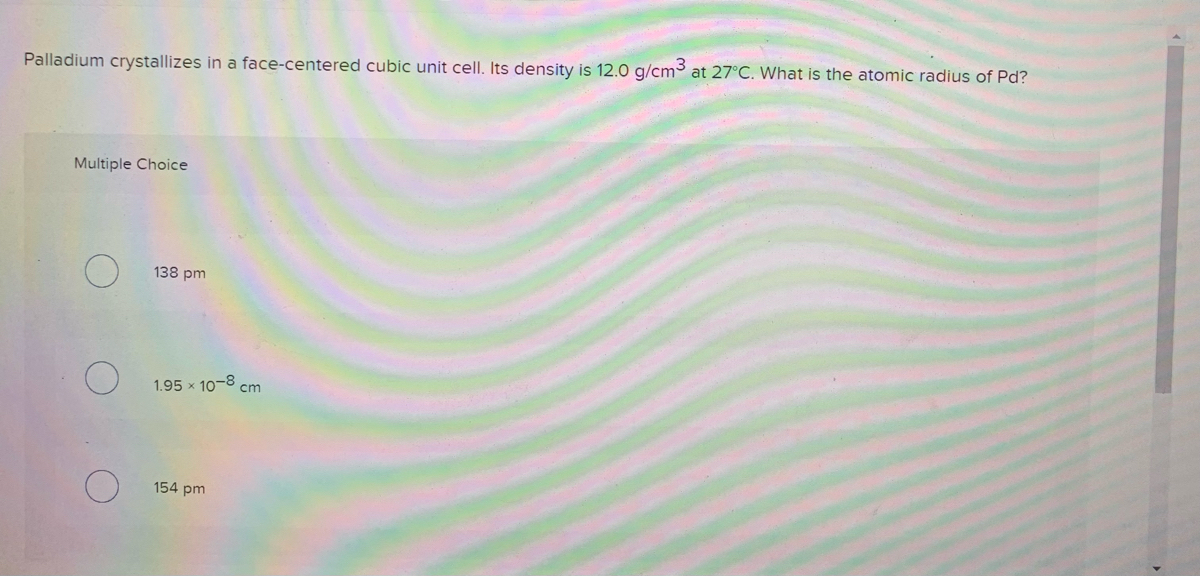
SOLVED: Palladium (at. wt. = 106) crystallizes in a face-centered cubic unit cell. Its density is 12.023 g/cm3 . Calculate the atomic radius of palladium and its packing efficiency.

SOLVED: A metal crystallizes in the face-centered cubic (FCC) lattice. The density of the metal is 8902 kglm , and the length of a unit cell edge, a,is 352.4 pm. Calculate the

SOLVED: Rhodium has a density of 12.41 g>cm3 and crystallizes with the face-centered cubic unit cell. Calculate the radius of a rhodium atom.

The metal palladium crystallizes in a face-centered cubic lattice with an edge length of 388.8 pm. What is the density of the palladium? | Homework.Study.com

Metallic lead crystallizes in a face-centred cubic lattice, with one Pb atom per lattice point. If the metallic radius of Pb is 175 pm, what is the volume of the unit cell

Document - Problem #1: Palladium crystallizes in a face-centered cubic unit cell. Its density is 12.023 g/cm3. Calculate the atomic radius of palladium. | Course Hero

Gold occurs as face centred cube and it has a density of 19.30 kg dm ^-3 .Calculate atomic radius of gold. (Molar mass of Au = 197 )